Instrumentation used for photochemical research
Diode-array spectrophotometer with optional external lamp
| In a diode-array spectrophotometer (like the one in our research group, a computer controlled (C) AnalytikJena SPECORD S600 (B)), a high intensity polychromatic light beam passes through the sample with enough light intensity to induce photochemical reactions (Ref. 1-3). The main adventage of the method is that following the reaction by sectrophotometry is obvious.
The induction of photochemical reaction is even more effective when using a high intensity external lamp. Our external lamp is either a Spectroline FC-100/F UV-A lamp emitting at 365 nm (A) or a 500 W NINGBO OALY halogen lamp (D). When used in the arrangement on the photo, they have a photon flux one order of magnitude higher as the photon flux from the internal lamps of the spectrophotometer in the whole (190-1100 nm) wavelength range (Ref. 4).
|
Top of page
Photochemical reactor combining a CCD spectrophotometer and a LED
| In the photochemical reactor made of brass (E), the photoreaction is induced from the top by a quasi-monochromatic LED irradiation source (A) and it is followed by an Avantes fibre-optic spectrophotometer (optical fibres: (F); spectrophotometer lamps: (C); CCD detector: (B)). The sample is placed into a normal, 1,000 cm pathlength cuvette (on the small Figure on the right-bottom, the reactor is opened so the cuvette can be seen). Because of the brass material of the reactor, the sample can be stirred with a magnetic stirrer (E).
The power supply (G) of the LED can be controlled by a computer making possible to use different illumination profiles. Also, there is the possibility of illuminating the sample independently of the detecting light beam (Ref. 1).
|
Top of page
Photochemical reactor combining a CCD spectrophotometer and an external lamp
| The photochemical reactor introduced in the previous paragraph can be used not only with LED but also with external Spectroline FC-100/F UV-A lamp radiation source (A) emitting at 365 nm, or a 500 W NINGBO OALY halogen lamp emitting in the visible wavelength range. The reaction is still followed by an Avantes fibre-optic spectrophotometer. Parts of the photoreactor and other complementaries (B-F) are the same as on the previous photo. |
Top of page
Automatic titrator with external lamp
When at least one of the products of a photochemical reaction is acid, then the kinetics of the acid formation can be followed by a continous back-titration of the acid formed in the process (e.g. by a pH-stat measurement, keeping the pH of the solution constant and adding the required amount of base to the solution). (Ref. 1: photochemical formation of hydroxy-quinones and hydroquinones (weak acids) from 1,4-benzoquinones). To follow the kinetics of the acid formation, a computer controlled automatic titrator system (C) is used. The illumination source is the Spectroline FC-100/F UV-A lamp emitting at 365 nm (A), or a 500 W NINGBO OALY halogen lamp emitting in the visible wavelength range. The reaction wessel (D) can be thermostatted. A base tube (E) and a pH-selective glass electrode (F) is immersed into the sample. To avoid carbonation, Ar gas (B) is used. The sample is mixed using a magnetic stirrer.
|
Top of page
Sample holder for measurements with light sensitive electrodes
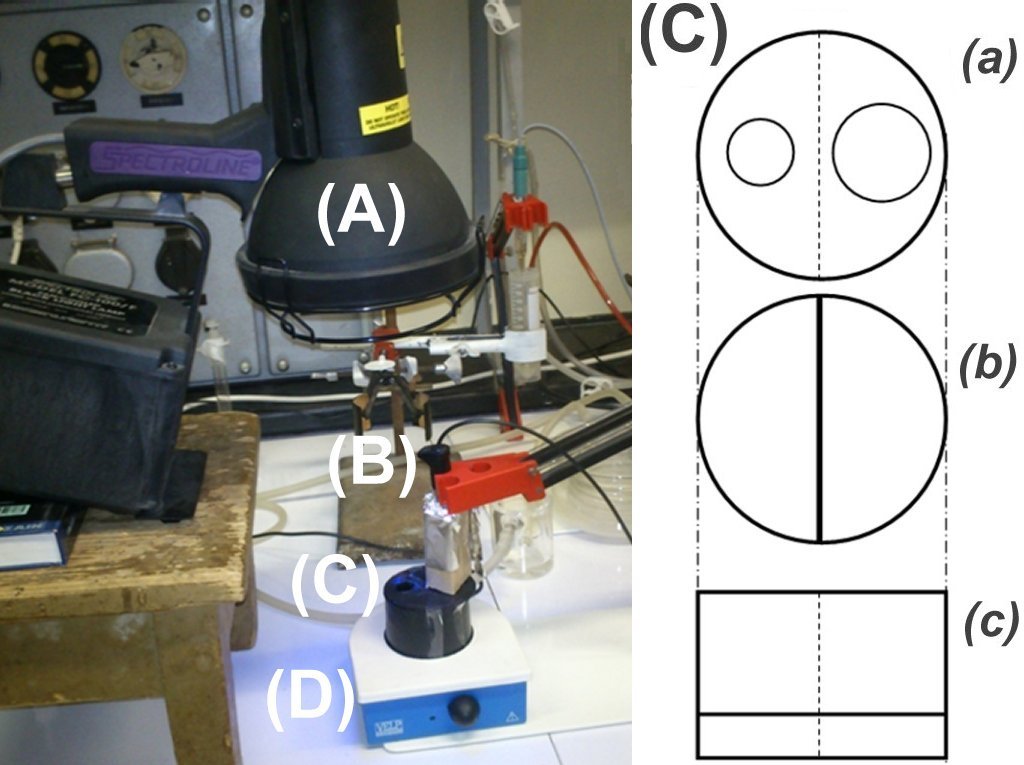
| Chloride ion selective electrodes (B) are light sensitive when irradiated by an intense UV or Vis light (e.g. a Spectroline FC-100/F UV-A lamp (A), or a 500 W NINGBO OALY halogen lamp). This problem can be eliminated by using a specially designed, stirred (D) and calibrated sample holder (C) which spatially separates the illumination and detection sites. The material of the photochemical sample holder (C) is black plastic (poly-propylene). The inside wall has a 10-mm high slit for the stir bar. The cap (a) has a 16-mm diameter whole for the electrode and a larger one for the UV light. The optimal volume of the sample holder is 50 mL. To record the electrode potential, a computer controlled automatic titrator system is used. (a) cap (top view) (b) sample holder (top view) (c) sample holder (side view) |
Top of page